
MS is applied to fields as disparate as airport security, food and wine analysis, drug and explosives analysis, as well as most fields of chemical and biological research. 1 (2) = 2 C 2 (2) = 4 H 1 (2) … cuartos en renta tijuana Overview Mass spectrometry (MS) is a proven analytical method used to glean information about the chemical structure of a chemical sample. Fragments containing both isotopes of Br can be seen in the mass spectrum of ethyl bromide:First, calculate the molar mass of the empirical formula: 1 (12 g/mole C) + 2 (1 g/mole H) + 1 (16 g/mole O) = 30 g/mole CH2O Next, Divide the molar mass of the substance by the molar mass of the empirical formula: 60/30 = 2 Finally, multiply the number of atoms of each element in the empirical formula by this ratio. macul n° 3354, macul, santiago de chileBecause there are two abundant isotopes of both chlorine (about 75% 35 Cl and 25% 37 Cl) and bromine (about 50% 79 Br and 50% 81 Br), chlorinated and brominated compounds have very large and recognizable M+2 peaks. Atomic mass of all elements (along with the rounded off values) is mentioned in the chart below. Examples can be found in the Nicolaou taxol total synthesis. The compound is used to protect alcohols in organic synthesis. It is a colorless or white solid that is soluble in many organic solvents but reacts with water and alcohols. The concentration of chloride in the blood is called serum chloride, and this concentration is regulated by the kidneys.It is a silane containing two methyl groups, a tert -butyl group, and a reactive chloride. coli and budding yeast are 10–200 mM (dependent on medium), in mammalian cells 5–100 mM and in blood plasma 100 mM. The one containing 37 Cl has a relative formula mass of 80 – hence the two lines at m/z = 78 and m/z = 80.Characteristic concentrations of chloride in model organisms are: in both E. The molecular ion containing the 35 Cl isotope has a relative formula mass of 78. The molecular ion peaks (M + and M+2) each contain one chlorine atom – but the chlorine can be either of the two chlorine isotopes, 35 Cl and 37 Cl. averagemass = (fractionof35Cl × massof 35Cl) + (fractionof 37Cl × massof 37Cl) If we let x represent the fraction that is 35 Cl, then the fraction that is 37 Cl is represented by 1.00 − x. 
The average mass of chlorine is the fraction that is 35 Cl times the mass of 35 Cl plus the fraction that is 37 Cl times the mass of 37 Cl.What is the percent composition of Cl in terms of these two isotopes? Show Answer Check Your Learning Naturally occurring chlorine consists of 35 Cl (mass 34.96885 amu) and 37 Cl (mass 36.96590 amu), with an average mass of 35.453 amu. It was discovered in 1770’s and soon became useful as a commercial agent. It is a chemical element that belongs to the halogen group with the symbol Cl. The atomic mass or relative isotopic mass refers to the mass of a single particle, …Chlorine is a green yellow gas with a very pungent odour that is twice as dense as air. all owner dealer Chlorine: Atomic number: 17: Atomic mass 35.453: Atomic mass 102: Density at STP 0.0032: Number of protons: 17: Number of neutrons (typical isotopes) 35 37: Number of electrons: 17: Electron configuration 3s2 3p5: Oxidation states +1,5,7/-1: Electron affinity 349: Electronegativity 3.16: First. What is the percent composition of Cl in terms of these two isotopes? Show Answer Check Your Learningrefresh results with search filters open search menu. 
The one containing 37 Cl has a relative formula mass of 80 – hence the two lines at m/z = 78 and m/z = 80. 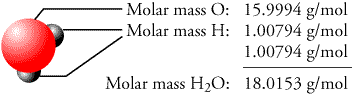
The molecular ion containing the 35 Cl isotope has a relative formula …The molecular ion peaks (M + and M+2) each contain one chlorine atom – but the chlorine can be either of the two chlorine isotopes, 35 Cl and 37 Cl. Cl mass The molecular ion peaks (M+ and M+2) each contain one chlorine atom - but the chlorine can be either of the two chlorine isotopes, 35 Cl and 37 Cl.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
